How can the DiCoDi® (Digital Cognitive Diagnostics) be used as a cognitive test in everyday practice?
Isabell Ballasch 1,2,3Marietta Meka 1
Jan-Patric Schmid 1
Elke Kalbe 2
Christian Vorstius 4
Josef Kessler 2,3
1 KOJ Hearing Network, Germany
2 Department of Medical Psychology | Neuropsychology & Gender Studies, Center for Neuropsychological Diagnostics and Intervention (CeNDI), Faculty of Medicine and University Hospital Cologne, University of Cologne, Germany
3 Department of Neurology, University of Cologne, Faculty of Medicine and University Hospital Cologne, Germany
4 Institute of Psychology, University of Wuppertal, Germany
Abstract
Hearing loss can be a potential risk factor for later dementia. At the same time, existing cognitive tests are mostly auditory-based and rely on intact sensory function, which limits their ability to accurately assess cognitive performance in individuals with hearing loss. Against this background, the Digital Cognitive Diagnostics (DiCoDi®) test was developed. DiCoDi® was evaluated for validity and reliability in 200 control participants and 173 individuals with hearing loss. Additionally, a comprehensive neuropsychological test battery was administered. As expected, all DiCoDi® subtests correlated significantly with their corresponding paper-and-pencil equivalents (p<0.001), while showing no significant association with a depression questionnaire (p>0.05). The test also demonstrated high test-retest reliability (p<0.0005). Accordingly, DiCoDi® is a valid and reliable tool for the early detection of cognitive deficits. It can be used alongside standard audiological diagnostics in ENT clinics and hearing aid centers, as well as by neurologists, neuropsychologists, and memory clinics.
Keywords
cognitive test, cognitive impairments, dementia, hearing loss, digital technology
Introduction
Increasing age is associated with a rise in sensory impairments [1]. Among those aged 60 to 69, nearly 20% can be classified as hearing impaired, while in the 70 to 79 age group, the proportion is about twice as high at 42%. In individuals aged 80 and above, around 72% are affected by hearing loss [2]. Besides hearing loss, cognitive impairments such as dementia also increase with advancing age. In 2021, approximately 1.8 million people in Germany were living with dementia [3].
Hearing loss in midlife is linked to an evaluated risk of developing dementia [4] and represents a potential risk factor for later dementia [5], [6]. Among 14 modifiable risk factors for dementia, hearing loss in midlife was identified as the most significant modifiable risk factor for future cognitive decline, alongside high LDL cholesterol [5]. Possible mechanisms linking hearing loss and dementia include a causal effect of hearing loss on cognition – either directly via reduced auditory input affecting cognitive brain regions, or indirectly through increased isolation, lower self-efficacy, reduced physical activity, or less cognitive engagement [1], [7], [8], [9]. Alternatively, cognitive decline may itself contribute to hearing impairment [10]. Although some studies suggest beneficial effects of hearing aid or cochlear implant use in this context, conclusive evidence is still lacking. Systematic reviews (e.g. [11], [12]) and large randomized controlled trials (e.g. [13]) highlight limitations in terms of generalizability. Furthermore many individuals delay or avoid using hearing aids due to stigma and perceived barriers, leading to a higher threshold for seeking compensation [14].
Moreover, the cognitive performance of individuals with hearing loss is often inadequately assessed, underestimated, or even misdiagnosed (false positives), as most of these cognitive tests are primarily auditory and assume an intact sensory system [15]. In suboptimal listening conditions – such as those caused by hearing loss – early speech processing requires greater cognitive effort, leaving fewer resources for encoding auditory information into memory [16]. Individuals with hearing loss tend to perform significantly worse on cognitive tests under unaided conditions, and the use of hearing amplification does not substantially improve test performance [17]. These findings highlight the need for cognitive assessments adapted for individuals with hearing loss, such as the HI-MoCA, MoCA-HI, MMSE for the hearing impaired, O-DEM, and DemTectEar. Due to limited validation and lack of German norms, only MoCA-HI and O-DEM are suitable for hearing-impaired individuals, though their complexity and paper-pencil-based format restricts use to specialized clinical settings and demands substantial resources. To overcome these limitations, digital tools such as the tablet-based CANTAB and the ALACog battery offer promising alternatives, though their use in routine care remains limited due to lengthy administration, required expertise, and the need for specialized training [18].
Against this background, the Digital Cognitive Diagnostics (DiCoDi®) was developed as a cognitive test for individuals with and without hearing loss. This short article aims to present the DiCoDi® and its application in everyday practice.
Method
DiCoDi®
The DiCoDi® is a digital cognitive test (see Figure 1 [Fig. 1]) consisting of nine subtests that cover a wide range of cognitive domains (see Table 1 [Tab. 1]). The subtests were selected based on the following criteria: a) comprehensive assessment of cognitive status, b) highest sensitivity within the cognitive domain, c) adaptability for tablet use, d) complete independence from hearing ability, and e) time efficiency and practical feasibility. The subtests were developed based on existing cognitive tests. The DiCoDi® takes 20 to 30 minutes to complete. Instructions and stimuli are presented visually (minimum font size 12). The participant operates the tablet independently, though the test administrator should be present to monitor the process. Evaluation is fully computer-based and automatic.
Figure 1: Example illustration of DiCoDi® subtests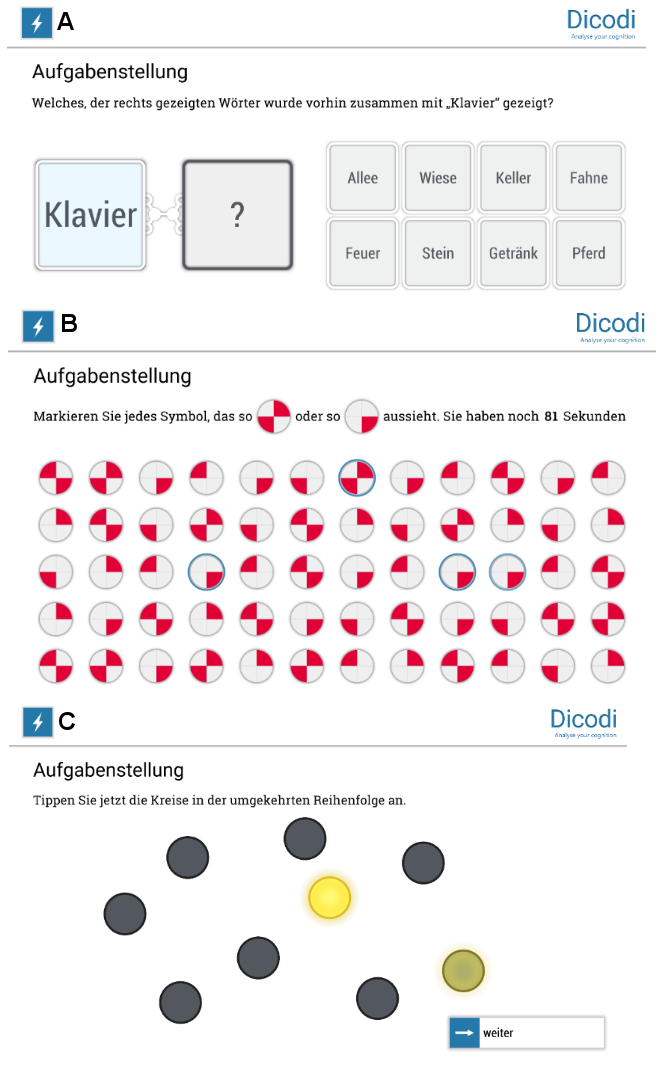
A: Remembering word pairs. B: Attention. C: Circle tapping task
(Figure available in German only)
Table 1: Overview of DiCoDi® subtests and their cognitive domains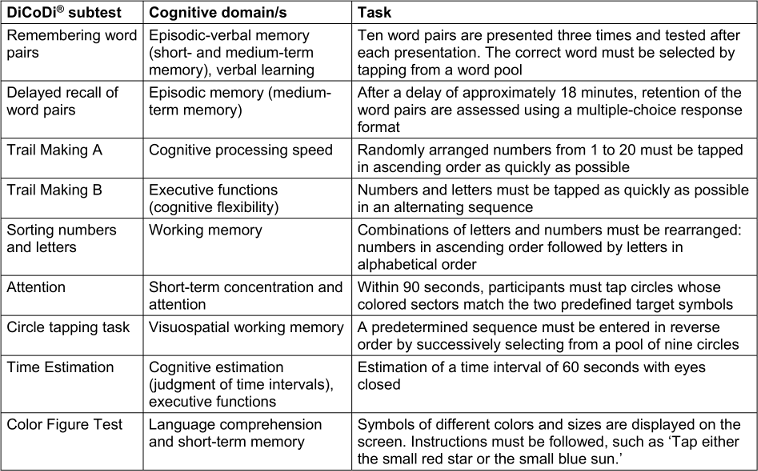
Sample and assessment
The DiCoDi® was evaluated in a sample of 373 individuals aged 40 years and older, including 200 participants in the control group (age: M=67.48, SD=12.2; women: n=116 [58.0%]) and 173 participants in the group with hearing loss (age: M=72.86, SD=8.7; women: n=81 [46.8%]) between 2022 and 2023. Hearing impairments in the control group were assessed based on self-reported history, four-point self-rating scale and the examiner’s evaluation. Individuals with hearing loss underwent ear-specific pure-tone audiometry at frequencies ranging from 250 to 8,000 Hertz. The mean hearing threshold was 44.06 dB (11.6) for the right ear, 42.89 dB (10.6) for the left ear, and 43.27 dB (10.2) averaged across both ears. According to the WHO the hearing loss group comprised 93 individuals with mild (53.9%), 77 with moderate (44.5%), 2 with severe (1.2%), and 1 with profound hearing loss or deafness (0.6%).
The control group was recruited by trained medical and psychology students in the greater Cologne area, Germany. Individuals with hearing loss were assessed in the hearing aid branches of the company KOJ in Switzerland by trained psychology students.
In addition to the DiCoDi®, a comprehensive neuropsychological test battery was administered to validate the DiCoDi® subtests.
Test-retest reliability of the DiCoDi® was examined in a separate control group (n=71; age: M=59.86, SD=9.5; women: n=40 [56.3%]), with a mean interval of nine weeks between assessments.
The study was approved by the ethical committee of the University of Wuppertal, Germany (reference number: MS/AE 220614), and was conducted in compliance with national laws and the 1975 Declaration of Helsinki. All participants gave their informed consent.
Statistical analysis
The statistical analyses were conducted using SPSS Statistics 29.0. Descriptive statistics, ANCOVAs with age and education as covariates, and two-tailed Spearman rank correlations were conducted.
Results
Table 2 [Tab. 2] shows the descriptive statistics and results of the ANCOVA for all DiCoDi® subtests. For subtests measured in seconds, the results include medians and interquartile ranges in addition to means and standard deviations. The control group and individuals with hearing loss demonstrated descriptively similar performances on the DiCoDi® subtests. ANCOVA results showed significant group differences in two subtests (Trail Making A and Time Estimation; p>0.001), with small to large effect sizes (pη²=0.05–0.24).
Table 2: Descriptive statistics and results of the ANCOVA for the DiCoDi® subtests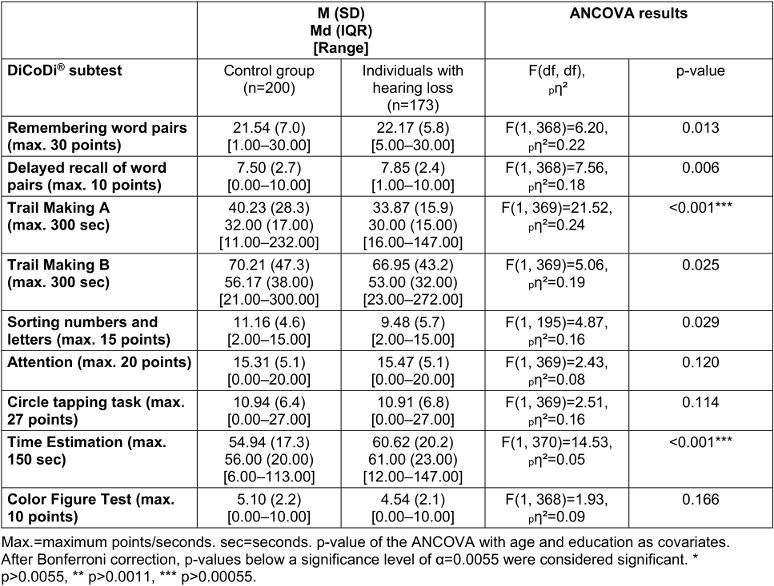
Regarding convergent validity, all DiCoDi® subtests showed a significant moderate to strong correlation with their respective paper-and-pencil equivalents (rs=0.24–0.67; p>0.001). Additionally, none of the subtests showed a significant correlation with Beck’s Depression Inventory V (p>0.05), demonstrating the divergent validity of the DiCoDi®.
Furthermore, all DiCoDi® subtests except the Color Figure Test showed significant correlations between the first and second test session, indicating high test-retest reliability (see Table 3 [Tab. 3]).
Table 3: Test-retest reliability of the DiCoDi® subtests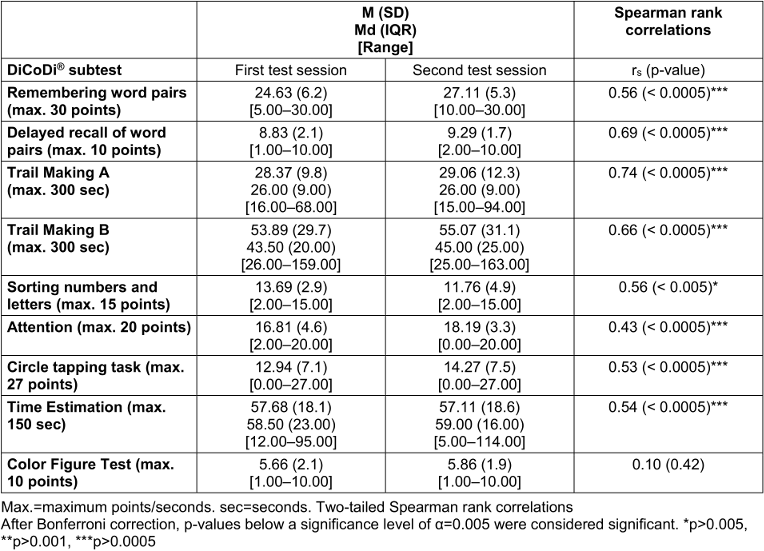
Discussion
The present study examined the applicability, reliability, and validity of the DiCoDi® in individuals with and without hearing loss. The results suggest that the DiCoDi® is a valid and reliable measure for assessing cognitive impairments in both populations.
The group comparison revealed similar performance between the control group and the hearing loss group across most DiCoDi® subtests. Interestingly, the hearing loss group showed slightly better performance than the control group on two DiCoDi® subtests: Trail Making A and Time Estimation. Regarding Time Estimation, both groups performed within the normative range for 1-minute estimation, as defined by Meyers et al. [19]. However, time estimation in individuals with hearing loss remains understudied, warranting further research. Additionally, a significant difference emerged between groups in the Trail Making A subtest, which primarily measures cognitive processing and visual scanning. To confirm the robustness of this effect, an ANCOVA with bootstrapping (5,000 resamples) was conducted. The bias-corrected and accelerated (BCa) confidence intervals indicated a significant effect, corroborating the initial finding. Nevertheless, descriptive statistics based on median and interquartile range suggested that the magnitude of this group difference was small, and a nonparametric Mann–Whitney-U test did not confirm a significant difference (p=0.117). These results therefore point to a modest yet statistically robust difference when age and education are controlled for. One possible explanation for the superior performance of the hearing loss group lies in their increased reliance on visual information. Individuals with hearing impairment often compensate for reduced auditory input through enhanced visual attention and faster visual processing. Supporting this, Campbell and Sharma [20] demonstrated cross-modal cortical reorganization in adults with mild-to-moderate hearing loss. Using electroencephalography (EEG), they found that hearing-impaired adults exhibited significantly larger visual evoked potential amplitudes and shorter latencies compared to normal-hearing controls. This indicates recruitment of auditory cortical areas for visual processing, reflecting adaptive cross-modal plasticity. Such cross-modal plasticity possibly underlies the subtle advantage observed in the visual scanning components of the Trail Making A task in the hearing loss group. Future studies should further investigate these findings.
As expected, the DiCoDi® subtests showed significant correlations with their paper-based counterparts and no associations with unrelated measures, supporting convergent and divergent validity. Further research is needed to compare the DiCoDi® with cognitive tests for individuals with hearing loss.
All DiCoDi® subtests except the Color Figure Test showed high test-retest reliability. The absence of a significant correlation in the Color Figure Test may result from low standard deviations despite highly similar mean scores across sessions, as minimal variability makes it harder to detect associations [21].
Application
The DiCoDi® can serve as a relevant diagnostic tool for the early detection of cognitive deficits in people aged 40 years and older. It can supplement routine audiological assessments in ENT practices and hearing aid centers and can be used by neurologists, neuropsychologists, and in memory clinics. Proper training is required to administer the DiCoDi®. When the DiCoDi® reveals indications of cognitive impairment, a comprehensive follow-up assessment is recommended. Suitable points of referral include general practitioners, neurologists, and memory clinics. If no signs of cognitive impairment are detected, it is advisable to repeat the test after approximately one year to monitor for any emerging subjective cognitive complaints. The DiCoDi® does not replace a medical diagnosis or a full neuropsychological evaluation.
Strenghts and limitations
A strength of this study is its large sample size, which enhances the generalizability of the findings. However, objective hearing data are lacking for the control group, as only self-assessments have been collected. Furthermore, most participants with hearing loss had only mild to moderate impairments, highlighting the need for further research on DiCoDi®’s applicability in more severe cases. The tablet-based, standardized administration and automated scoring make the DiCoDi® especially suitable for routine use in resource-limited clinical settings by minimizing examiner bias.
Conclusion
The DiCoDi® is a reliable and valid digital cognitive test for individuals with and without hearing loss. It is intended to improve the early detection of cognitive impairments in ENT settings. While it does not replace a full neuropsychological test battery, it can offer preliminary insights into possible cognitive deficits. Future research should examine the psychometric property of the DiCoDi® and its applicability in diverse clinical populations, although the large and representative sample suggests a high degree of data stability.
Notes
Conference presentation
This contribution was presented at the 27th Annual Conference of the German Society of Audiology and published as an abstract [22].
Competing interests
Prof. Dr. Josef Kessler and Prof. Dr. Elke Kalbe developed the DiCoDi® in cooperation with the KOJ Hearing Network, Germany, which provided the tablets, audiometric assessments, software programming, and facilities for data collection in the hearing-impaired group. Furthermore, the KOJ Hearing Network is handeling the distribution of the DiCoDi®. Isabell Ballasch and Marietta Meka are part-time employees of KOJ Hearing Network. Jan-Patric Schmid is the Managing Director of KOJ Hearing Network.
References
[1] Völter C, Thomas JP, Maetzler W, Guthoff R, Grunwald M, Hummel T. Sensory Dysfunction in Old Age. Dtsch Arztebl Int. 2021 Jul 26;118(29-30):512-20. DOI: 10.3238/arztebl.m2021.0212[2] von Gablenz P, Holube I. Prävalenz von Schwerhörigkeit im Nordwesten Deutschlands : Ergebnisse einer epidemiologischen Untersuchung zum Hörstatus (HÖRSTAT) [Prevalence of hearing impairment in northwestern Germany. Results of an epidemiological study on hearing status (HÖRSTAT)]. HNO. 2015 Mar;63(3):195-214. DOI: 10.1007/s00106-014-2949-7
[3] Blotenberg I, Hoffmann W, Thyrian JR. Dementia in Germany: Epidemiology and Prevention Potential. Dtsch Arztebl Int. 2023 Jul 10;120(27-28):470-6. DOI: 10.3238/arztebl.m2023.0100
[4] Cantuaria ML, Pedersen ER, Waldorff FB, Wermuth L, Pedersen KM, Poulsen AH, Raaschou-Nielsen O, Sørensen M, Schmidt JH. Hearing Loss, Hearing Aid Use, and Risk of Dementia in Older Adults. JAMA Otolaryngol Head Neck Surg. 2024 Feb 1;150(2):157-64. DOI: 10.1001/jamaoto.2023.3509
[5] Livingston G, Huntley J, Liu KY, Costafreda SG, Selbæk G, Alladi S, Ames D, Banerjee S, Burns A, Brayne C, Fox NC, Ferri CP, Gitlin LN, Howard R, Kales HC, Kivimäki M, Larson EB, Nakasujja N, Rockwood K, Samus Q, Shirai K, Singh-Manoux A, Schneider LS, Walsh S, Yao Y, Sommerlad A, Mukadam N. Dementia prevention, intervention, and care: 2024 report of the Lancet standing Commission. Lancet. 2024 Aug 10;404(10452):572-628. DOI: 10.1016/S0140-6736(24)01296-0
[6] Yu RC, Proctor D, Soni J, Pikett L, Livingston G, Lewis G, Schilder A, Bamiou D, Mandavia R, Omar R, Pavlou M, Lin F, Goman AM, Gonzalez SC. Adult-onset hearing loss and incident cognitive impairment and dementia - A systematic review and meta-analysis of cohort studies. Ageing Res Rev. 2024 Jul;98:102346. DOI: 10.1016/j.arr.2024.102346
[7] Griffiths TD, Lad M, Kumar S, Holmes E, McMurray B, Maguire EA, Billig AJ, Sedley W. How Can Hearing Loss Cause Dementia? Neuron. 2020 Nov 11;108(3):401-12. DOI: 10.1016/j.neuron.2020.08.003
[8] Uchida Y, Sugiura S, Nishita Y, Saji N, Sone M, Ueda H. Age-related hearing loss and cognitive decline - The potential mechanisms linking the two. Auris Nasus Larynx. 2019 Feb;46(1):1-9. DOI: 10.1016/j.anl.2018.08.010
[9] Wayne RV, Johnsrude IS. A review of causal mechanisms underlying the link between age-related hearing loss and cognitive decline. Ageing Res Rev. 2015 Sep;23(Pt B):154-66. DOI: 10.1016/j.arr.2015.06.002
[10] Maharani A, Dawes P, Nazroo J, Tampubolon G, Pendleton N; SENSE-Cog WP1 group. Trajectories of recall memory as predictive of hearing impairment: A longitudinal cohort study. PLoS One. 2020 Jun 18;15(6):e0234623. DOI: 10.1371/journal.pone.0234623
[11] Carasek N, Lamounier P, Maldi IG, Bernardes MND, Ramos HVL, Costa CC, Bahmad F Jr. Is there benefit from the use of cochlear implants and hearing aids in cognition for older adults? A systematic review. Front Epidemiol. 2022 Aug 30;2:934750. DOI: 10.3389/fepid.2022.934750
[12] Yeo BSY, Song HJJMD, Toh EMS, Ng LS, Ho CSH, Ho R, Merchant RA, Tan BKJ, Loh WS. Association of Hearing Aids and Cochlear Implants With Cognitive Decline and Dementia: A Systematic Review and Meta-analysis. JAMA Neurol. 2023 Feb 1;80(2):134-41. DOI: 10.1001/jamaneurol.2022.4427
[13] Lin FR, Pike JR, Albert MS, Arnold M, Burgard S, Chisolm T, Couper D, Deal JA, Goman AM, Glynn NW, Gmelin T, Gravens-Mueller L, Hayden KM, Huang AR, Knopman D, Mitchell CM, Mosley T, Pankow JS, Reed NS, Sanchez V, Schrack JA, Windham BG, Coresh J; ACHIEVE Collaborative Research Group. Hearing intervention versus health education control to reduce cognitive decline in older adults with hearing loss in the USA (ACHIEVE): a multicentre, randomised controlled trial. Lancet. 2023 Sep 2;402(10404):786-97. DOI: 10.1016/S0140-6736(23)01406-X
[14] Marcos-Alonso S, Almeida-Ayerve CN, Monopoli-Roca C, Coronel-Touma GS, Pacheco-López S, Peña-Navarro P, Serradilla-López JM, Sánchez-Gómez H, Pardal-Refoyo JL, Batuecas-Caletrío Á. Factors Impacting the Use or Rejection of Hearing Aids-A Systematic Review and Meta-Analysis. J Clin Med. 2023 Jun 13;12(12):4030. DOI: 10.3390/jcm12124030
[15] Kim MW, Jin MH, Choi JY, Kwak MY. Potential overestimation of cognitive impairment because of hearing loss: impact of test modalities on cognitive test scores. J Laryngol Otol. 2023 Aug;137(8):845-50. DOI: 10.1017/S0022215123000154
[16] Füllgrabe C. On the Possible Overestimation of Cognitive Decline: The Impact of Age-Related Hearing Loss on Cognitive-Test Performance. Front Neurosci. 2020 Jun 9;14:454. DOI: 10.3389/fnins.2020.00454
[17] Saunders GH, Odgear I, Cosgrove A, Frederick MT. Impact of Hearing Loss and Amplification on Performance on a Cognitive Screening Test. J Am Acad Audiol. 2018 Jul/Aug;29(7):648-55. DOI: 10.3766/jaaa.17044
[18] Ferguson MA, Nakano K, Jayakody DMP. Clinical Assessment Tools for the Detection of Cognitive Impairment and Hearing Loss in the Ageing Population: A Scoping Review. Clin Interv Aging. 2023 Dec 7;18:2041-51. DOI: 10.2147/CIA.S409114
[19] Meyers JE. Time estimation: Close your eyes and tell me when a minute goes by. Appl Neuropsychol Adult. 2020 May-Jun;27(3):279-83. DOI: 10.1080/23279095.2018.1535435
[20] Campbell J, Sharma A. Cross-modal re-organization in adults with early stage hearing loss. PLoS One. 2014 Feb 28;9(2):e90594. DOI: 10.1371/journal.pone.0090594
[21] Maxwell SE, Delaney HD, Kelley K. Designing experiments and analyzing data: A model comparison perspective. 3rd ed. New York: Routledge; 2017.
[22] Ballasch I, Meka M, Schmid JP, Kalbe E, Vorstius C, Kessler J. Wie kann der DiCoDi (Digital Cognitive Diagnostic) als kognitiver Test im Praxisalltag eingesetzt werden? In: Deutsche Gesellschaft für Audiologie e. V.; ADANO, editors. 27. Jahrestagung der Deutschen Gesellschaft für Audiologie und Arbeitstagung der Arbeitsgemeinschaft Deutschsprachiger Audiologen, Neurootologen und Otologen. Göttingen, 19.-21.03.2025. Düsseldorf: German Medical Science GMS Publishing House; 2025. Doc032. DOI: 10.3205/25dga032




